Neuroscience Confirms Julian Jaynes’s Neurological Model
Posted June 10, 2020. Updated March 18, 2024.
Beginning in 1999, research began to emerge confirming Julian Jaynes’s neurological model for the bicameral mind: fMRI studies showing a right/left temporal lobe interaction during auditory verbal hallucinations.
Yet, more than twenty years later, there remains ongoing confusion on this subject (for example, a recent post on Scott Alexander’s “Star Slate Codex” blog was completely wrong on this issue). To help clear up this lingering confusion, I revisit this topic with an excerpt from one of my related newsletter articles, followed by quotes from related research, related video, further reading, and additional resources:
“Over three decades ago, Julian Jaynes introduced his theory of the origin of consciousness and a historically older mentality called the bicameral mind …
While the technology was not then available to test his ideas, Jaynes suggested a possible neurological model for the bicameral mind. Briefly stated, he reasoned that auditory hallucinations emanate from the areas of the right temporal lobe corresponding to the language areas in the left temporal lobe, and are subsequently processed (or “heard”) in the left temporal lobe language areas. Because of their external quality, the bicameral person experiencing auditory hallucinations interpreted them as the externally generated commands of a dead ancestor, chief, king, or god. To this day, those who experience auditory hallucinations often hear behavioral commands and experience them as being externally generated.
In my chapter in Reflections on the Dawn of Consciousness, “Consciousness, Hallucinations, and the Bicameral Mind: Three Decades of New Research,” I discuss a number of neuroimaging studies that emerged over the past decade that provide support for Jaynes’s neurological model. These studies show brain activation in the right followed by the left temporal lobe at the onset of auditory hallucinations. Some researchers infer a possible interaction between these two areas — the language areas of the left hemisphere and the corresponding areas of the right hemisphere — during auditory hallucinations.
Since the publication of Reflections on the Dawn of Consciousness, a number of new studies have been published that also show bilateral temporal lobe (‘bicameral’) activation during auditory hallucinations, providing further support for Jaynes’s neurological model specifically and his bicameral mind theory in general.
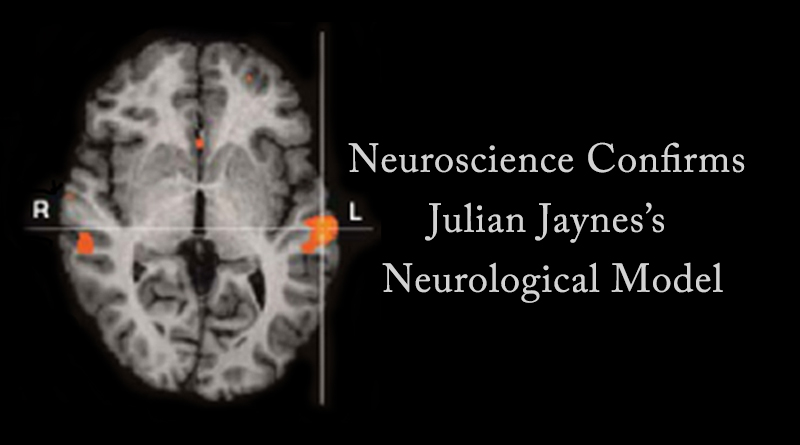
In 2007, French psychiatrist Renaud Jardri and his colleagues published a study in Molecular Psychiatry titled “Activation of Bilateral Auditory Cortex during Verbal Hallucinations in a Child with Schizophrenia.” They used functional MRI to study the “neural substrates of verbal auditory hallucinations in a child suffering from very early onset schizophrenia.” The analysis performed “shows bilateral activation of the superior temporal gyri” during auditory hallucinations (see the image above). This bilateral activation of the superior temporal gyri during auditory hallucinations is exactly what Jaynes predicted more than 30 years ago.
In an article published in 2007 in Schizophrenia Research, Dr. Iris E. C. Sommer and her colleagues in the Netherlands reported that “the majority of schizophrenia patients showed prominent activity in the right-sided homologues of the classical language areas during AVH [auditory verbal hallucinations] (i.e. in the right inferior frontal gyrus, right superior temporal and supramarginal gyrus), while normal language is generally produced in the left hemisphere in right-handed subjects.” Again, this corresponds precisely with Jaynes’s predictions.
In another study published in 2008, Sommer et al. measured cerebral activation using fMRI in 24 psychotic patients. They report that “group analysis for AVH revealed activation in the right homologue of Broca’s area, bilateral insula, bilateral supramarginal gyri and right superior temporal gyrus.” They go on to speculate that “the association between AVH and activity in right hemisphere language areas could explain the low linguistic complexity and derogatory content, characteristic for AVH in psychotic patients.”
In a 2008 article titled “Functional MRI of Verbal Self-Monitoring in Schizophrenia” published in Schizophrenia Bulletin, psychologist Veena Kumari and her colleagues in the United Kingdom conclude that positive schizophrenic symptoms (hallucinations and persecution) show “exaggerated activation in the right superior-middle temporal gyrus.”
Also noteworthy, Dr. Yair Lampl in the Department of Neurology at Tel Aviv University and his colleagues published an article in late 2005 that studied auditory hallucinations in stroke patients. According to Lampl, auditory hallucinations occur only rarely after a stroke. Of the 641 stroke patients in the study, only four experienced auditory hallucinations. Of interest to the present discussion, all of the cases of auditory hallucinations “occurred after an ischemic lesion of the right temporal lobe.”
These new studies provide additional evidence in support of Jaynes’s neurological model. They have been published in peer-reviewed journals by a variety of researchers working in different labs around the world. While their research is highly supportive of Jaynes’s neurological model, these scientists did not have the goal of providing evidence for Jaynes’s theory and in some cases may not even be aware of it. …”
Reprinted from Marcel Kuijsten, “New Evidence for Jaynes’s Neurological Model: A Research Update,” The Jaynesian, Volume 3, Issue 1. Read the full article.
Statements Supportive of Jaynes’s Neurological Model by Psychiatrists, Neurologists, and Neurosurgeons
“Neuroimaging techniques of today have illuminated and confirmed the importance of Jaynes’ hypothesis.”
— Robert Olin, M.D., Ph.D., Professor Emeritus in Preventive Medicine, Karolinska Institute, Stockholm, Sweden, in Lancet, 1999, 20, 354, 9173.
“In 1976 Julian Jaynes proposed in his book … [that] the left hemisphere was the site for speech, and the right hemisphere was the site for hallucinations that expressed voices and commands of gods and demons. … Their results provided direct evidence of the involvement of primary auditory areas in auditory hallucinations. … Contemporary neuroimaging data have been used to revive and support … [Jaynes’s] controversial hypothesis.“
— Leo Sher, M.D., Journal of Psychiatry and Neuroscience, 2000, 25, 3, 239-40.
“… Scientific interest in [Jaynes’s] work has been re-awakened by the consistent findings of right-sided activation patterns in the brain, as retrieved with the aid of neuroimaging studies in individuals with verbal auditory hallucinations.”
— Jan Dirk Blom, M.D., in A Dictionary of Hallucinations (Springer, 2009).
“An interesting hypothesis is posed by Julian Jaynes on the “bicameral brain” (sic) where one hemisphere (right) gives orders and the other (left) listens and executes orders. … The latest studies have shown bilateral activation of Broca’s area instead of only left side activation that brings new life to the theory of mind bicameralism. The reduction of lateralization even correlated with the intensity of hallucinations.“
— D.M. Pavlović, M.D., A.M. Pavlović, M.D., and Maja Lačković, M.D., “The Neuropsychology of Hallucinations,” Archives of Biological Sciences, 2011, 63, 1, 43-48.
“Jaynes claims that remnants of the ancient bicameral brain (sic) organization can still be found in the verbal or auditory hallucinations associated with hypnosis and schizophrenia. This hypothesis is consistent with the findings from recent neuroimaging studies, which identified the right temporal lobe as the source of auditory hallucinations in patients with schizophrenia.“
— Andrea E. Cavanna and Andrea Nani, Consciousness: Theories in Neuroscience and Philosophy of Mind (Springer, 2014).
“The theory of a bicameral mind evolved the psychology and neuropsychiatric concepts of some disorders including the one for schizophrenia. … Jaynes’ theories were further confirmed by … other authors. … According to all these findings and theories, it is postulated that anterior corpus callosotomy would be beneficial in controlling the auditory and visual hallucinations in those with schizophrenia refractory to the highest medical therapy.“
— Mousa Taghipour, MD & Fariborz Ghaffarpasand, MD, “Corpus Callosotomy for Drug-Resistant Schizophrenia; Novel Treatment Based on Pathophysiology,” World Neurosurgery, August 2018, 116: 483-484.
Supportive Evidence from Peer-Reviewed Articles
The Neurology of Auditory Hallucinations: Evidence from Direct Stimulation, EEG, PET , & fMRI
Key Point: Numerous studies using a variety of techniques implicate the language areas of the non-dominant hemisphere in auditory verbal hallucinations.
“Electrical stimulation of the temporal lobe was performed in chronic schizophrenic patients to treat the auditory hallucinations. During electrical stimulation, there appeared different kinds of hallucinations. … In most of these cases, craniotomy of the non-dominant side was performed, and the exposed temporal lobe cortex was electrically stimulated directly.”
— Ishibashi, et al., “Hallucinations Produced by Electrical Stimulation of the Temporal Lobes in Schizophrenic Patients,” Journal of Experimental Medicine, 1964, 82, 124-139.
“Penfield reproduced such experiences by electrical stimulation of the temporal lobe cortex. We have found that similar hallucinatory experiences may arise from subcortical stimulation of the temporal lobe.”
— M. J. Horowitz and J. E. Adams, “Hallucinations on Brain Stimulation: Evidence for Revision of the Penfield Hypothesis,” in W. Keup (ed.), Origin and Mechanisms of Hallucinations (Springer, 1970).
“In reviewing our series of patients it was of interest to note that hallucinations were produced by electrical stimulation of the depth structures of the temporal lobe in certain patients.“
— Weingarten, et al. ,”The Relationship of Hallucinations to the Depth Structures of the Temporal Lobe,” Acta Neurochirurgica, 1977, Suppl. 24: “Advances in Stereotactic and Functional Neurosurgery 2”, 199-216.
“Metabolism in Broca’s region and its right-hemisphere homologue correlated positively and significantly in the hallucinating group, as it did in anterior cingulate and left superior temporal areas, and in right frontal and parietal areas.”
— Cleghorn, et al., “Regional Brain Metabolism During Auditory Hallucinations in Chronic Schizophrenia,” British Journal of Psychiatry, 1990, 157, 562-570.
“Left temporal lobe activity was found to be decreased with increasing positive symptom’s scores scores. … Patients with a recent history of auditory hallucinations showed an atypical right temporal lobe dominance, which occurred independently of medication status.”
— Gordon, et al., “Single Photon Emission Computed Tomography (SPECT) Measures of Brain Function in Schizophrenia,” Australian and New Zealand Journal of Psychiatry, 1994, 28, 446–452.
“A large and significant decrease in SSVEP latency in the right temporo/parietal region occurred in the second prior to the report of auditory hallucinations. … This finding suggests that activity of fine temporal resolution in the neural networks in the right temporo/parietal area may be implicated in the genesis of auditory hallucination. …”
— Line, et al., “Steady State Visually Evoked Potential Correlates of Auditory Hallucinations in Schizophrenia,” NeuroImage, 1998, 8, 4.
“Metabolism in Broca’s region and its right-hemisphere homologue correlated positively and significantly in the hallucinating group…”
— Cleghorn, et al., “Regional Brain Metabolism During Auditory Hallucinations in Chronic Schizophrenia,” The British Journal of Psychiatry, 1990, 157, 4, 562-570.
“Results showed that activation [during auditory hallucinations] first appeared in the right middle temporal gyrus and then extended to a wider area of the right superior temporal and left superior temporal gyri (where the hallucination is “heard”), right middle and inferior frontal gyri, right anterior cingulate, and right cuncus.”
—- Lennox, et al., “Spatial and Temporal Mapping of Neural Activity Associated with Auditory Hallucinations,” Lancet, 1999, 353, 644.
“The group of hallucinating patients demonstrated right medial and inferior frontal activations (BA 45/46), focal bilateral temporal lobe activations (BA 22, 37), and a left parahippocampal gyral activation.”
— Copolov, et al., “A PET Study of Brain Activation in Response to Auditory Hallucinations and External Speech in Schizophrenic Patients,” Biological Psychiatry, April 2000, 47, 8.
“Patients showed no differences while generating inner speech but experienced a relatively attenuated response in the posterior cerebellar cortex, hippocampi, and lenticular nuclei bilaterally and the right thalamus, middle and superior temporal cortex, and left nucleus accumbens during auditory verbal imagery.”
— Shergill, et al, “Functional Anatomy of Auditory Verbal Imagery in Schizophrenic Patients With Auditory Hallucinations,” American Journal of Psychiatry, October 2000, 157, 1691–1693.
“Auditory hallucinations were associated with activation in the … temporal cortex bilaterally (with greater responses on the right).“
— Shergill, et al, “Mapping Auditory Hallucinations in Schizophrenia Using Functional Magnetic Resonance Imaging,” The Archives of General Psychiatry, November 2000, 57.
“Group analysis demonstrated shared areas of activation in right and left superior temporal gyri, left inferior parietal cortex and left middle frontal gyrus.“
— Lennox, et al., “The Functional Anatomy of Auditory Hallucinations in Schizophrenia,” Psychiatry Research, November 2000, 100, 1, 13–20.
“AVHs [auditory verbal hallucinations] were associated with increased metabolic activity in the left primary auditory cortex and the right middle temporal gyrus. Our results suggest a possible interaction between these areas during AVHs.”
— Bentaleb, et al., “Cerebral Activity Associated with Auditory Verbal Hallucinations: A Functional Magnetic Resonance Imaging Case Study,” Journal of Psychiatry & Neuroscience, 2002, 27, 2, 110.
“Recent functional neuroimaging findings seem to confirm the hypothesis that the right middle temporal gyrus represents the source of auditory hallucinations in at least some schizophrenic patients. Arguably, this lateralization pattern could well be the reason why these inner voices lack the characteristic of being self-generated.“
— Cavanna et al. “The ‘Bicameral Mind’ 30 Years On,” Functional Neurology, 2007, 22, 1, 11-15.
“The majority of schizophrenia patients showed prominent activity in the right-sided homologues of the classical language areas during AVH [auditory verbal hallucinations] (i.e. in the right inferior frontal gyrus, right superior temporal gyrus and supramarginal gyrus), while normal language is generally produced in the left hemisphere in right-handed subjects.”
— Sommer, et al., “Can fMRI-guidance Improve the Efficacy of rTMS Treatment for Auditory Verbal Hallucinations?” Schizophrenia Research, 2007, 93, 1, 406–8.
“Group analysis for AVH revealed activation in the right homologue of Broca’s area, bilateral insula, bilateral supramarginal gyri and right superior temporal gyrus. … The association between AVH and activity in right hemisphere language areas could explain the low linguistic complexity and derogatory content, characteristic for AVH in psychotic patients.“
— Sommer, et al., “Auditory Verbal Hallucinations Predominantly Activate the Right Inferior Frontal Area,” Brain, 2008, 131, 3169-77.
“The regions (Broca’s homologue and the right insula) coincide with the areas that showed greatest activation during auditory verbal hallucinations in our analysis of 24 patients (Sommer et al., 2008). It could therefore be hypothesized that auditory verbal hallucinations result from ‘release’ language activity in the right inferior frontal area that is inhibited in the healthy brain.”
— Sommer & Diederen, “Language Production in the Non-Dominant Hemisphere as a Potential Source of Auditory Verbal Hallucinations,” Brain, 2009, 132, 10, e124.
“… Group analysis for AVH [auditory verbal hallucinations] revealed activation in the right homolog of Broca’s area, bilateral insula, bilateral supramarginal gyri, [and] right superior temporal gyrus.”
— Sommer, Iris & Kelly Diederen, “Auditory Verbal Hallucinations and Language Lateralization,” in Iris E.C. Sommer and René S. Kahn (eds.), Language Lateralization and Psychosis (Cambridge University Press, 2009).
“Patients experiencing AVHs demonstrated significantly increased activation likelihoods in a bilateral neural network, including the Broca’s area…”
— Jardri, et al., “Cortical Activations During Auditory Verbal Hallucinations in Schizophrenia: A Coordinate-Based Meta-Analysis,” The American Journal of Psychiatry, 2011, 168, 1, 73–81.
“Findings suggest that heightened functional coupling between the left inferior frontal gyrus and right temporal regions leads to coactivation in these speech processing regions that is hallucinogenic.”
—- Hoffman, et al., “Time Course of Regional Brain Activity Accompanying Auditory Verbal Hallucinations in Schizophrenia,” The British Journal of Psychiatry, 2011, 198, 4, 277–283.
“Auditory verbal hallucinations are a cardinal symptom of schizophrenia … The current results indicate that spatial location of auditory hallucinations is associated with the right temporoparietal junction (rTPJ) anatomy, a key region of the ‘where’ auditory pathway.”
— Plaze, et al., “Where Do Auditory Hallucinations Come From?” A Brain Morphometry Study of Schizophrenia Patients with Inner or Outer Space Hallucinations,” Schizophrenia Bulletin, January 2011, 37, 1, 212-21.
“Several common areas of activation were observed for the psychotic and nonpsychotic subjects during the experience of AVH, consisting of the bilateral inferior frontal gyri, insula, superior temporal gyri, supramarginal gyri and postcentral gyri, left precentral gyrus, inferior parietal lobule, superior temporal pole, and right cerebellum.”
— Diederen, et al., “Auditory Hallucinations Elicit Similar Brain Activation in Psychotic and Nonpsychotic Individuals,” Schizophrenia Bulletin, September 2012, 38, 5.
“AVH [auditory verbal hallucination]-related activity was … observed in Broca’s area and its right hemisphere homologue…”
— Zmigrod, et al., “The Neural Mechanisms of Hallucinations: A Quantitative Meta-Analysis of Neuroimaging Studies,” Neuroscience & Biobehavioral Reviews, 2016, 69, 113-123.
“AVHs [auditory verbal hallucinations] were associated with activation in bilateral language and motor regions.“
— Ford, et al., “Neurobiology of Auditory Hallucinations,” Oxford Research Encyclopedia of Neuroscience, November 2019.
“The group with complex hallucinations showed elevated activity in the bilateral temporal cortex including Wernicke’s area…”
— Marschall, et al., “Spontaneous Brain Activity Underlying Auditory Hallucinations in the Hearing-Impaired,” Cortex, March 2021, 136, 1-13.
“We [found] activations … during experience of AVH: these involved Wernicke’s area and its right homologue, Broca’s area and its right homologue, and the precentral gyrus and supplementary motor area bilaterally.”
— Fuentes-Claramonte, et al., “Auditory hallucinations activate language and verbal short-term memory, but not auditory, brain regions,” Scientific Reports, 2021, 11, 18890.
Language Lateralization and Psychosis
Key Point: Individuals that process language more evenly in both hemispheres (“less lateralized”) are more likely to experience auditory hallucinations.
“Delay in establishing dominance in one hemisphere could be the critical factor that predisposes to schizophrenia.”
— Crow, T.J., “Cerebral Lateralization is Delayed in Children who Later Develop Schizophrenia,” Schizophrenia Research, 1996, 22, 3, 181–185.
“These results show the strong association of the right middle temporal gyrus with the experience of auditory hallucination in this patient, supporting the hypothesis that auditory hallucinations reflect abnormal activation of auditory cortex. Language is more associated with the left hemisphere in normal subjects. This finding may therefore reflect the abnormal lateralisation of language function in schizophrenia.“
—- Lennox, et al., “Spatial and Temporal Mapping of Neural Activity Associated with Auditory Hallucinations,” Lancet, 1999, 353, 644.
“Twelve schizophrenic patients and twelve healthy controls were scanned while they were engaged in a verb-generation and a semantic decision task. … The results indicate that language processing is less lateralized in patients than in controls. … Analysis of variance of the extent of activity … revealed a significant hemisphere by group interaction … which was due to increased activation in the right hemisphere of the patients.“
— Sommer, et al., “Language Lateralization in Schizophrenia, An fMRI Study,” Schizophrenia Research, 2001, 52, 1-2, 57-67.
“Schizophrenia patients showed reduced language lateralization in the frontal cortex, because of a more bilateral activation of Broca’s area compared with a primarily left hemisphere activation in healthy controls.“
— Weiss, et al., “Language Lateralization in Unmedicated Patients During An Acute Episode of Schizophrenia: A functional MRI Study,” Psychiatry Research, March 2006, 31, 146.
“Both the overall auditory cortex and planum temporale volumes and the lateralization to the left hemisphere were markedly reduced in patients. The decrease of lateralization correlated with increased severity of symptoms.
— Oertel, et al., “Reduced Laterality as a Trait Marker of Schizophrenia—Evidence from Structural and Functional Neuroimaging,” The Journal of Neuroscience, February 2010, 30, 6, 2289–2299.
“Reduced cerebral lateralisation of language in patients diagnosed with schizophrenia has been documented in a substantial number of studies.“
— Allen and Modinos, “Structural Neuroimaging in Psychotic Patients with Auditory Verbal Hallucinations,” in Blom and Sommer (eds.), Hallucinations: Research and Practice (Springer, 2012).
“…[R]educed language lateralization is a weak trait marker for schizophrenia as such and a strong trait marker for the experience of auditory hallucinations within the schizophrenia population”
— Ocklenburg , et al. “Auditory Hallucinations and Reduced Language Lateralization in Schizophrenia: A Meta-Analysis of Dichotic Listening Studies,” Journal of the International Neuropsychological Society, April 2013, 19, 4, 410-8.
“Patients with schizophrenia exhibited significantly decreased leftward hemispheric [language] lateralization.”
— Alary, et al., “Functional Hemispheric Lateralization for Language in Patients with Schizophrenia,” Schizophrenia Research, Sept. 2013, 149, 1-3, 42-7.
Right Hemisphere as “Alien Intruder”/Sensed Presence/Externally Perceived Voice
Key Point: The origin of auditory hallucinations in the non-dominant hemisphere may be what gives them their “external” quality.
“In schizophrenia, defective interhemispheric integration, probably neurochemical, may lead to disinhibition of the awareness by the left hemisphere that it is being “influenced” by an unknown “external force” which is fact the right hemisphere.”
— Nasrallah, Henry A., “The Unintegrated Right Cerebral Hemispheric Consciousness as Alien Intruder: A Possible Mechanism for Schneiderian Delusions in Schizophrenia?” Comprehensive Psychiatry, 1985, 26, 3, 273–282.
“The hypothesis of vectorial hemisphericity predicts that left hemispheric intrusions of the right hemispheric equivalent of the sense of self should be associated with the experience of a “presence” of someone else. The neurophenomenological profile of a woman whose medical history satisfied these theoretical criteria … is presented. ”
— Persinger, et al., “The Sensed Presence as Right Hemispheric Intrusions into the Left Hemispheric Awareness of Self: An Illustrative Case Study,” Perceptual and Motor Skills, 1994, 78, 3.
“We report evidence of isolated conceptual knowledge in the right hemisphere of a woman with chronic anarchic hand sign after ischemic infarction of the central four-fifths of the corpus callosum.”
— Baynes, et al., “Isolation of a Right Hemisphere Cognitive System in a Patient with Anarchic (Alien) Hand Sign,” Neuropsychologia, 1997, 35, 8.
“The second component is alienation, which is the failure to recognize the content of AVH as self-generated. This failure may be related to the fact that cerebral activity associated with AVH is predominantly present in the speech production area of the right hemisphere.”
— Sommer, et al., “Dissecting Auditory Verbal Hallucinations into Two Components: Audibility (Gedankenlautwerden) and Alienation (Thought Insertion),” Psychopathology, 2010, 43, 137–140.
“[A] lack of synchronization between Broca and its homolog may lead to the erroneous interpretation of emotional speech activity from the right hemisphere as coming from an external source.”
— Ćurčić-Blake, et al., “When Broca Goes Uninformed: Reduced Information Flow to Broca’s Area in Schizophrenia Patients With Auditory Hallucinations,” Schizophrenia Bulletin, September 2013, 39, 5.
Alien hand syndrome after corpus callosotomy reveals its underlying mechanism. 3 out of a series of 18 patients demonstrated ALS after callosotomy. Distinct features were bihemispheric language dominance and genuine left-handedness. Both hemispheres come with capacities for independent action. Loss of contralateral inhibition appears a major mechanism underlying ALS.
— Helmstaedter, et al., “Dr. Strangelove Demystified: Disconnection of Hand and Language Dominance Explains Alien-Hand Syndrome after Corpus Callosotomy,” Seizure, 2021, 86.
Auditory and Visual Hallucinations and Temporal Lobe Epilepsy
Key Point: Patients with temporal lobe epilepsy (abnormal brain activity in the temporal lobes) also occasionally experience both auditory and visual hallucinations.
See Section 2, “Auditory Hallucinations in Temporal Lobe Epilepsy” (Case Reports)
— Shafica Karagulla, “Psychical Phenomena in Temporal Lobe Epilepsy and The Psychoses,” British Medical Journal, March 1955, 1, 4916, 748–752.
“A case report is presented to illustrate problems in managing the schizophrenia-like psychosis of temporal lobe epilepsy.”
— Sidney Bloch, “Problems of Treatment in the Schizophrenia-Like Psychosis of Temporal Lobe Epilepsy-A Case Report,” Australian and New Zealand Journal of Psychiatry, 1968, 2.
“One of them — a 32-year-old female with a right temporo-occipital focus — had a seizure pattern which included a vision of Christ coming down from the sky.”
— Dewhurt and Beard, “Sudden Religious Conversions in Temporal Lobe Epilepsy,” British Journal of Psychiatry, 1970, 117, 540, 497-507.
“Hallucinations are infrequently seen in prepubertal children. … We here report the case of a child who presented with psychosis and nocturnal hallucinations and for whom temporal epilepsy was eventually diagnosed and successfully treated.”
— Kechid, et al., “Hearing Hallucinations in a 12-Year-Old Child: Psychotic Disorders or Temporal Epilepsy?”, The Primary Care Companion to the Journal of Clinical Psychiatry, 2008, 10, 4, 328–329.
“MEG revealed interictal epileptic discharges within the anterior partition of the right superior temporal gyrus; signal-to-noise ratio of these discharges was overall poor in EEG. The findings suggest that auditory vocal hallucinations without verbal content can evolve in the right hemisphere and are probably independent of language lateralization.“
— Hug, et al., “Voices Behind the Left Shoulder: Two Patients with Right-Sided Temporal Lobe Epilepsy,” Journal of the Neurological Sciences, June 2011, 305, 1–2.
“The psychoses of epilepsy are well recognized complications of seizure disorders, diagnosed easily from the history. However, in the absence of recognized seizures, the diagnosis can be challenging.”
— Edward Needham, “Temporal Lobe Epilepsy Masquerading As Psychosis – A Case Report and Literature Review,” Neurocase: The Neural Basis of Cognition, 2012, 18, 5.
“Hearing voices (i.e. auditory verbal hallucinations) is mainly known as part of schizophrenia and other psychotic disorders. However, hearing voices is a symptom that can occur in many psychiatric, neurological and general medical conditions. … The third patient is a 27-year-old woman with voices caused by temporal lobe epilepsy.“
— I.E. Sommer and D.W. van der Spek, “Hearing Voices Does Not Always Constitute A Psychosis,” Nederlands Tijdschrift Voor Geneeskunde, 2016, 160, D492.
“The purpose of this case report is to document a patient with cinematographic hallucinations, with corresponding right temporal lobe seizures on electroencephalogram (EEG).“
— Nelson, et al., “A Unique Patient with Epilepsy with Cinematographic Visual Hallucinations,” Epilepsy & Behavior Case Reports, 2016, 5, 78-79.
“We therefore propose that the mediation of AVH in the context of schizophrenia spectrum disorders involves the attribution of an excess of negative salience by anterior-cingulate areas to linguistic input from Broca’s right homologue.”
— J. Looijestijn, et al., “Draining the pond and catching the fish: Uncovering the ecosystem of auditory verbal hallucinations,” NeuroImage: Clinical, 2018, 20.
“Activation during VAH [verbal auditory hallucination] experience was observed … The left and right inferior frontal gyri (Broca’s area and its contralateral homologue) were involved.” … “Specifically, our findings show that miscommunications between two functional networks are central in the occurrence of hallucinations, i.e. linguistic content from the right-sided area of Broca is assigned false salience in the insula.”
— J. Looijestijn, et al., Exploring Function in the Hallucinating Brain, University of Groningen, 2018.
“Psychiatric disorders are very common in epilepsy. … Here, we report a case of a young female who was suffering from complex partial seizure with secondary generalization and who subsequently developed schizophrenic symptoms. … the patient who had a right-sided lesion developed schizophrenic symptoms.”
— B. Sweetha, “A Case of Untreated Mesial Temporal Sclerosis in Right Hemisphere Presenting As Schizophrenia,” University Journal of Medicine and Medical Specialities, 2019.
Further Evidence from Split-Brain Experiments
Psychological experiments conducted with “split-brain” patients, who have had their corpus callosum severed (the primary connection between the two brain hemispheres), provide further evidence for Jaynes’s neurological model. These experiments suggest that the two hemispheres can in some ways operate independently, express different preferences, that one’s sense of self is associated with the language-dominant hemisphere, and that the actions of the right hemisphere can often feel “alien” to the person (with the right hand holding a newspaper to read and the left hand knocking it away, for example). See Elizabeth Schechter, Self-Consciousness and “Split” Brains: The Minds’ I (Oxford University Press, 2018) for a recent, extensive discussion of this fascinating research.
Related Video
Iris Sommer: Investigating Voices
Describes the experience and neurology of auditory hallucinations and shows the accuracy of Julian Jaynes’s neurological model.
2-Minute Neuroscience: Functional Magnetic Resonance Imaging (fMRI)
Explains the basics of fMRI as well as some of its limitations.
How does fMRI brain scanning work? Alan Alda and Dr. Nancy Kanwisher, MIT
During the filming of “Brains on Trial with Alan Alda,” Alan made a special stop in a mock fMRI scanner to understand how it works and how scientists can glean insights.
Further Reading
Julian Jaynes, “The Double Brain,” in J. Jaynes, The Origin of Consciousness in the Breakdown of the Bicameral Mind (Houghton Mifflin, 1976/1990), Ch. I.5.
Marcel Kuijsten, “Consciousness, Hallucinations, and the Bicameral Mind: Three Decades of New Research,” in M. Kuijsten (ed.), Reflections on the Dawn of Consciousness (Julian Jaynes Society, 2006).
Robert Olin, “Auditory Hallucinations and the Bicameral Mind,” Lancet, 1999, 354, 166.
Leo Sher, “Neuroimaging, Auditory Hallucinations, and The Bicameral Mind,” Journal of Psychiatry and Neuroscience, 2000, 25, 3, 239-40.
Elizabeth Schechter, Self-Consciousness and “Split” Brains: The Minds’ I (Oxford University Press, 2018).
Iris Sommer and Rene S. Kahn (eds.), Language Lateralization and Psychosis (Cambridge University Press, 2009).
Mousa Taghipour and Fariborz Ghaffarpasand, “Corpus Callosotomy for Drug-Resistant Schizophrenia; Novel Treatment Based on Pathophysiology,” World Neurosurgery, August 2018,116, 483-484.
Review additional peer-reviewed supporting evidence for Julian Jaynes’s neurological model.
Brain fMRI image reprinted by permission from Macmillan Publishers Ltd: Jardri, et al, “Activation of Bilateral Auditory Cortex during Verbal Hallucinations in a Child with Schizophrenia,” Molecular Psychiatry, 12, 319, Copyright 2007.
Learn more about Julian Jaynes’s theory by joining the Julian Jaynes Society and reading our books.